NASAL VESTIBULE — The nasal vestibule is
lined with squamous epithelium. As such, tumors arising there are
most often squamous cell carcinomas (SCCA) and have a natural
history similar to that of squamous cell skin cancer. Primary tumors
of the nasal vestibule are usually relatively limited at
presentation and exhibit a more favorable prognosis than tumors of
the nasal cavity.
Overview — The nasal vestibule is a
pear-shaped cavity which functions as the entrance to the nasal
cavity. Its borders are the nasal septum and columella medially, the
lower lateral (alar) cartilage laterally, and the pre-maxilla
inferiorly. The vestibule terminates posteriorly at the limen nasi,
the junction of the lower and upper lateral cartilages, as well as
the transition from skin to mucosa. The vestibule is lined by skin
bearing hair follicles, sebaceous glands, and sweat glands.
The male-to-female ratio of cancer of the
nasal vestibule is 2:1, with an average age of 60 to 65 years. The
development of nasal vestibule tumors has been associated with
smoking tobacco. Although the majority of nasal vestibule tumors are
squamous cell carcinomas, basal cell carcinomas are also seen
Early tumors present as superficial
ulcerations or masses of the membranous septum or columella with
crusting, scabbing, and occasional minor bleeding. More advanced
lesions can perforate the septum or invade the alar cartilage and
grow through the skin surface. There can also be extention
inferiorly into the gingivo-labial sulcus and premaxillary soft
tissues. Growth into the posterior nasal cavity occurs late or with
recurrences.
The nasal vestibule drains primarily to the
submandibular lymph nodes. The facial lymph nodes are second-echelon
nodes, which may become involved with recurrent disease.
Only about 5 to 10 percent
of patients present with lymph node metastasis
History and physical exam, computed tomography
(CT) scan, and biopsy for pathologic confirmation are usually
sufficient for evaluation
Treatment — The goal of therapy is to
achieve the best oncologic outcome with the least morbidity in terms
of cosmesis and nasal function.
For small T1 lesions, either surgical excision or definitive
radiation therapy (RT) can be used. Large T1, T2, T3, and small T4
tumors are best treated with definitive RT, as surgical
excision of these lesions usually produces significant cosmetic
deformity. Extensive T4 tumors, however, require a combined modality
approach consisting of both surgery and radiation therapy.
Because of the rare occurrence of nasal
vestibule tumors, there are no prospective data to guide treatment
recommendations; the majority of the literature is
single-institution case series.
Early stage — T1 and T2 tumors are treated
with definitive RT, with a
five-year local control rate of 95 percent for T1 and T2 tumors and
for all tumor stages a five-year cause-specific survival and overall
survival of 90 and 76 percent, respectively. Surgery is a
reasonable alternative if the lesion can be removed with a good
functional and cosmetic result.
Definitive RT usually involves external beam
RT (EBRT) followed by a boost, delivered via EBRT or interstitial
brachytherapy one to two weeks after completing EBRT. EBRT can be
delivered either with opposed lateral fields using megavoltage
photons or a single anterior portal using a 4:1 mix of high-energy
electrons and megavoltage photons. For both techniques, a
tissue-equivalent wax bolus is placed over the entire nose to
convert the irregular nasal surface to a box-like contour to improve
the dose distribution. The dose delivered by the initial EBRT fields
is usually 50 gray (Gy) given in once-daily 2 Gy fractions. This is
followed by a boost to the tumor of 20 to 25 Gy. Serious
complications with definitive RT are uncommon.
Although interstitial implants of the nasal
vestibule are individualized, the implant generally consists of two
to four planes of needles inserted through the dorsum of the nose.
The source is iridium-192. When given as a boost after EBRT, the
dose delivered by the interstitial implant is usually 25 Gy. Very
early T1 tumors can be treated with interstitial brachytherapy
alone, usually 55 to 75 Gy, depending on the tumor size
Surgical resection of early stage lesions in
the nasal vestibule results in local control rates of 90 to 100
percent. Surgery can be a simple wide local excision with or without
skin grafting, but can also involve the removal of cartilage
(usually either columella or alar cartilage) and/or external nasal
skin.
It is important that the surgeon assess the
likelihood of achieving a complete resection; a lesion that is
likely to result in close or positive margins with surgery would be
better treated with definitive RT. The surgeon should also
critically assess the tumor extent to understand which structures
will need to be resected before embarking on the procedure. Nasal
floor or lateral nasal wall tumors can be easily removed if there is
not deep invasion into the nasal cartilages. Septal lesions can be
removed with excellent cosmesis as long as the anterior septal
support is not compromised. If needed, the external nose can be
reconstructed but often requires multiple procedures by a nasal
reconstructive surgeon and may be disfiguring.
Advanced stage — T3 and small T4 tumors with
no bone invasion are usually treated with RT alone, with surgery
reserved for radiation failure. RT is delivered as described above,
with EBRT to about 50 Gy followed by an interstitial brachytherapy
or EBRT boost for an additional 25 Gy.
Extensive T4 tumors should be treated with a
combination of surgery and RT, as these tumors have a low rate of
local control with either modality alone [10].
In this case, when the treatment consists of both surgery and RT, RT
can be delivered either pre- or postoperatively. Preoperative RT
offers the possibility of shrinking the tumor to allow for a less
extensive resection; postoperative RT results in less wound
complications. The EBRT dose is usually 50 to 60 Gy delivered in 2
Gy fractions.
Resection of advanced stage lesions may
require total rhinectomy. A prosthesis is usually created to cover
the defect, as total nasal reconstruction is cosmetically
unacceptable.
Little has been published about the use of
chemotherapy in the treatment of nasal vestibule cancers.
Management of the neck — Since the risk of
cervical metastasis is low, elective nodal treatment (either
radiation or neck dissection) should be considered only for patients
with T4 cancers, particularly those that are poorly differentiated
and/or recurrent. Patients who present with lymph node-positive
disease should be treated with radiation to the involved neck and/or
neck dissection.
Nodal recurrence rates of 0 to 13 percent have
been reported, with the majority of nodal failures occurring in
patients with stage T4 and/or recurrent tumors Furthermore, salvage
treatment of nodal disease is often successful. As an example, 75
patients with NO disease treated without elective neck irradiation
achieved an initial nodal control rate of 87 percent and a five-year
nodal control rate of 97 percent after salvage attempts
Prognosis — Five-year
local control is 80 to 95 percent for early-stage lesions and 70
percent for advanced lesions. Reported five-year overall
survival ranges from 58 to 76 percent. Regional and distant
metastases are extremely rare. Factors associated with a worse
prognosis include higher T stage, nodal involvement, and involvement
of the lip, cartilage, or bone
NASAL CAVITY — Tumors
arising in the nasal cavity, posterior to the limen nasi, have a
natural history similar to tumors of the paranasal sinuses and are
treated similarly.
Overview — Nasal cavity tumors are twice as
common in men and usually occur between the ages of 55 and 65 years.
Tobacco smoke has been associated with the development nasal cavity
tumors Adenocarcinomas of the nasal cavity have been linked to
multiple occupational exposures such as wood dust, formaldehyde,
leather dust, and textile dust
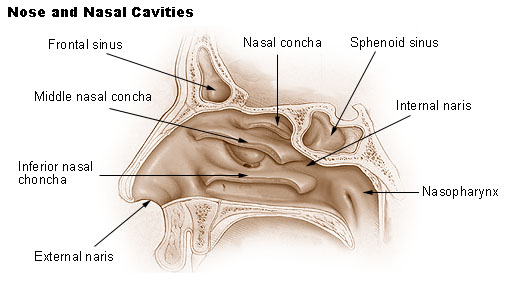
The nasal cavity begins at the limen nasi and
ends posteriorly at the choanae, which communicate directly with the
nasopharynx. It is bounded inferiorly by the hard palate and
superiorly by the base of skull. It is divided into right and left
halves by the nasal septum. The lateral wall of each nasal cavity is
composed of thin bony folds that project into the nasal cavity,
called the inferior, middle, and superior nasal turbinates (concha).
The nasal cavity communicates with the paranasal sinuses and other
structures via multiple passages or meatuses
Histology — Cancers with a wide variety of
histologies arise in the nasal cavity; these are similar to those
that occur in the paranasal sinuses.
- Squamous cell carcinoma (SCCA) is the
most common, accounting for 38 to 66 percent of cases
- Minor salivary gland tumors account for
30 percent of neoplasms in the nasal cavity. Adenocarcinoma is
the most common histology, followed by adenoid cystic, benign
mixed, and mucoepidermoid carcinoma Acinic cell carcinoma
is rare in the nasal cavity and paranasal sinuses.
- Neuroendocrine carcinomas consist of a
spectrum of rare histologies that include esthesioneuroblastoma
(olfactory neuroblastoma), sinonasal undifferentiated carcinoma
(SNUC), small cell carcinoma, and neuroendocrine carcinoma
- Inverted papilloma is a rare, benign
neoplasm that arises in the Schneiderian epithelium that lines
the nasal cavity. It is associated with squamous cell carcinoma
in approximately 5 to 10 percent of patients and is locally
aggressive, even in the absence of squamous cell carcinoma
- Other neoplasms that occur in the nasal
cavity include lymphomas, sarcomas, and malignant melanoma
Patients with tumors of the nasal cavity
usually present with a chronic history of sinus congestion and
recurrent nasal obstruction. Many patients complain of nasal
discharge and intermittent epistaxis. These symptoms can mimic those
of chronic sinusitis or nasal polyps and may delay the diagnosis of
cancer. However, since chronic sinusitis is most often associated
with bilateral symptoms, unilateral symptoms should be used as an
indication for further evaluation, such as nasal endoscopy and/or
imaging.
The symptoms from more advanced lesions depend
on the site of tumor spread: widening of the bridge of the nose from
lesions invading the olfactory region, frontal headaches from
extension through the cribriform plate or into the ethmoid sinus,
and proptosis, diplopia, or a palpable orbital mass from invasion of
the medial orbit.
The nasal cavity drains to the
retropharyngeal, submandibular, junctional, and subdigastric nodes.
Less than 10 percent of
patients present with lymph node metastasis
A detailed history and physical examination
should be performed with attention to signs and symptoms of orbital
or cranial nerve involvement. Biopsy can usually be performed via
nasal endoscopy. Both computed tomography (CT) with contrast and
magnetic resonance imaging (MRI) are usually performed, CT for
determining bone, nasopharyngeal, and lymph node invasion and MRI
for evaluating intracranial, orbital, and perineural spread. MRI
also helps determine whether the sinuses are opacified due to tumor,
fluid, or secondary inflammatory mucosal changes
Treatment — Both early and moderately
advanced tumors of the nasal cavity are
treated with a combination
of surgical resection and postoperative radiation. Very
advanced (unresectable tumors) are treated with RT with or without
concurrent chemotherapy.
No randomized studies have evaluated treatment
options for patients with carcinoma of the nasal cavity. Most
retrospective studies combine nasal cavity tumors with paranasal
sinus tumors, making it difficult to make conclusions that are
specific to nasal cavity cancers.
Early stage — For T1N0 tumors, surgery alone
is a reasonable treatment option, resulting in an overall survival
of 50 to 70 percent in selected patients However, most patients with
early stage tumors are treated with surgery and postoperative RT,
details of which are discussed below
Advanced stage — The standard treatment for
advanced stage tumors is surgery followed by postoperative RT.
Although preoperative RT has been used to minimize tumor bulk and
limit the extent of resection, we favor postoperative RT to minimize
the risk of wound complications. Very advanced (unresectable) tumors
are treated with definitive RT. Based upon experience with analogous
tumors of the sinus and other mucosal sites, chemotherapy,
administered as induction or concurrently with RT, is used by some
for unresectable and more extensive lesions, but there is little
supporting data.
Local control following surgery alone is 50 to
60 percent at five years. The addition of postoperative radiotherapy
improves five-year local control to 55 to 80 percent
The most common surgical techniques used for
exposure of the nasal cavity are the lateral rhinotomy and the
midface degloving procedures. The goal of both surgical techniques
is to provide an en bloc resection of all involved bone and soft
tissue, while preserving function and cosmesis. If the superior
extent of the tumor involves the fovea ethmoidalis or cribriform
plate, then a craniofacial resection is required
Endoscopic resection is gaining acceptance as
an alternative to transfacial or craniofacial surgeries
There is no consensus on criteria used to
define tumors as unresectable. Most agree that tumors that invade
the cavernous sinus or optic chiasm or extend transdurally are not
resectable without causing unacceptable morbidity and loss of
function
RT is initiated four to six weeks after
surgery to allow for healing. A dose of 60 Gy delivered in 2 Gy
fractions daily is sufficient when negative margins are achieved. If
surgery resulted in positive margins or gross residual disease, or
if the tumor was removed piecemeal (ie, endoscopically), the dose
used is 66 to 70 Gy in 2 Gy daily fractions. Hyperfractionated
irradiation may decrease the risk of optic neuropathy if any part of
the optic apparatus is likely to receive doses in excess of the
tolerance dose
RT for nasal cavity cancer is almost always
delivered using intensity-modulated radiation therapy (IMRT) rather
than conventional EBRT. IMRT allows for improved target coverage
while better sparing critical structures such as the optic
apparatus. Included in the target volume are the tumor bed, any
residual disease, and areas with a high likelihood of harboring
microscopic disease. Preoperative diagnostic CT, MRI and/or positron
emission topography (PET) scans are used when delineating the target
volume. Low rates of late complications, including blindness, have
been reported with IMRT
Very advanced or unresectable tumors are
treated with high doses of RT, usually 70 Gy in 2 Gy daily fractions
or 74.4 Gy in 1.2 Gy fractions delivered twice daily with a six-hour
split. Outcomes in these patients are poor, with five-year overall
survival of 15 percent reported Induction chemotherapy and/or
concurrent chemotherapy is being adapted for use in nasal cavity and
paranasal sinus cancers, despite scant evidence of chemotherapy
efficacy in nasal cavity cancer
Management of the neck — Elective
treatment of the N0 neck is controversial. Reported rates of nodal
recurrence when the neck is not electively treated vary from 0 to 19
percent Some believe that there is no benefit to
electively treating the N0 neck . Others recommend elective nodal
treatment, either neck dissection or radiation, for patients with
advanced-stage disease and/or poor prognostic factors. We suggest
elective irradiation of the N0 neck in patients with T3 and T4
disease, tumor extension to areas rich in lymphatics (ie, the
nasopharynx), and all esthesioneuroblastoma and sinonasal
undifferentiated carcinomas (SNUC).
Patients who present with lymph node-positive
disease should be treated with neck dissection and postoperative
radiation.
Prognosis — Local failure is problematic in
the treatment of nasal cavity tumors and adversely impacts survival;
regional and distant metastases are rare. When treated with surgery
and adjuvant RT, the five-year local control and overall survival is
50 to 80 percent and 50 to 70 percent, respectively. Factors
associated with a worse prognosis include higher T stage, treatment
with RT alone, lymph node involvement, SCCA and SNUC histologies,
and invasion of the orbit, cribriform plate, or dura
POSTTREATMENT SURVEILLANCE — Regular
posttreatment follow-up has become an essential part of the care of
patients after potentially curative treatment of head and neck
cancer (HNC). It is particularly important for nasal vestibule
cancers because recurrences can often be salvaged successfully.
Patients should be educated about possible signs and symptoms of
tumor recurrence, including hoarseness, pain, dysphagia, bleeding,
and enlarged lymph nodes.
In general, the intensity of follow-up is
greatest in the first two to four years, since approximately 80 to
90 percent of all recurrences after curative intent treatment will
occur within this timeframe. Continued follow-up beyond five years
is generally suggested since the risk of recurrence remains elevated
beyond the first five years, especially for cancers of the
hypopharynx, larynx, nasopharynx, and salivary glands and for second
primary malignancies. Because of the higher risk of recurrence and
second primary malignancy in those who continue tobacco use, many
schedule more frequent surveillance visits for these patients and
continue for longer duration (ie, beyond five years).
Posttreatment surveillance of head and neck
cancer, including screening for treatment-related complications, is
reviewed separately.
SUMMARY AND RECOMMENDATIONS
Nasal vestibule — Carcinomas of the nasal
vestibule are essentially skin cancers and have a good prognosis.
- We suggest that patients with early-stage
(T1-T3) tumors receive primary treatment with RT alone. Surgical
excision is an option if the lesion can be removed with a good
functional and cosmetic result.
- We suggest that patients with T4 tumors
involving bone undergo surgical resection followed by adjuvant
RT
- We suggest prophylactic nodal irradiation
only for patients with T4 cancers, particularly those that are
poorly differentiated and/or recurrent
Nasal cavity — Tumors arising in the nasal
cavity have a natural history similar to tumors of the paranasal
sinuses. A wide variety of tumor histologies occur in the nasal
cavity, with squamous cell carcinoma and adenocarcinoma
predominating.
- Patients with early and advanced stage
disease are treated with the combination of surgical resection
and adjuvant RT. We suggest postoperative RT, which minimizes
wound complications, rather than preoperative RT. For T1N0
tumors, surgery alone is a reasonable treatment option.
- We suggest prophylactic nodal irradiation
for patients with stage T3-T4 disease, tumor extension to areas
rich in lymphatics (ie, nasopharynx), and any
esthesioneuroblastoma or sinonasal undifferentiated carcinoma
(SNUC)
- For very advanced tumors, we suggest
combined modality therapy with chemotherapy added to radiation,
as induction and/or concurrent chemoradiation.