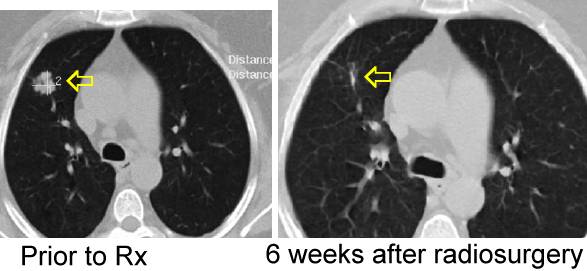
 |
Radiosurgery - using
high dose, very targeted radiation may be effective in sterilizing lung
cancer more effectively than conventional dose radiation using one single
high dose fraction (as below) or a few fractions (go
here,
here, and here).
It may be necessary to implant fiducials to target the beam (go here). The CT images can be used to estimate the volume of the cancer (see here).
|
Clinical
outcomes of single-fraction stereotactic radiation therapy of lung tumors
Ryusuke Hara, Cancer 2006;106:1347
The objective of the current study was to investigate the effects and the morbidities of single-fraction stereotactic radiation therapy (SRT) for lung tumors. Recently, wide applications have been identified for stereotactic radiation therapy (SRT) in the treatment of solid tumors, especially lung and liver tumors. In localized tumors, favorable results comparable to those achieved with surgical intervention were obtained with hypofractionated SRT. From the standpoint of normal tissue tolerance, single, high-dose irradiation is prohibitive. With the advent of SRT, it has been demonstrated that single, high-dose irradiation is tolerable if the planning target volume (PTV) is very small and if radiation therapy is delivered by 3-dimensional (3-D), converging, noncoplanar arcs. This single-fraction, high-dose radiation therapy is administered mainly to intracranial tumors. Therefore, it presumably is possible to extrapolate that the same single-fraction, high-dose irradiation can be delivered to lung tumors provided that the PTV is small enough and that radiation therapy is delivered by 3-D, noncoplanar, converging beams. However, to our knowledge, there are only a few reports to date regarding the use of SRT with a single fraction, and the optimal single-fraction dose for controlling local tumor remains to be established.
We have developed a Microtron-based system
for SRT of solid tumors that delivers radiation at a predetermined
respiratory phase. Patients with lung tumors have been treated by a
single-fraction SRT using this system since August 1998. In this
article, we report the outcomes of their treatment. The macroscopic tumor
volume (GTV)[ ranged from 1 cc to 19 cc (mean, 5 cc). In this study,
we assumed that the clinical target volume (CTV) was equal to the GTV
displayed by CT images of the lung field windows. For the PTV, 5-mm margins
were taken into account in all directions from the CTV. Before
December 2001, the prescribed dose was calculated by using the minimal dose
of the GTV = CTV as a reference target volume (29 tumors). Thereafter, the
prescribed dose was calculated as the minimal dose to the PTV (30 tumors).
Nine tumors received the minimal dose of 20 grays (Gy) or 25 Gy, and 50
tumors received ![]() 30 Gy (range, 30-34 Gy).
30 Gy (range, 30-34 Gy).
A Microtron device was modified to deliver
stereotactic irradiation under respiratory gating. Between August 1998 and
December 2004, 59 malignant lung tumors (11 primary tumors, 48 metastases)
that measured < 40 mm in greatest dimension were treated by single-fraction
SRT. Nine tumors received a minimal dose of < 30 grays (Gy), and 50 tumors
received a minimal dose of ![]() 30 Gy. The
macroscopic target volume ranged from 1 cc to 19 cc (mean, 5 cc).
30 Gy. The
macroscopic target volume ranged from 1 cc to 19 cc (mean, 5 cc).
The
1-year and 2-year local progression-free rates (LPFRs) were 93% and 78%,
respectively. The overall survival rate was 76.5% at 1 year and 41% at 2
years. Local regrowth of the irradiated tumor was a direct cause of
death in two patients. Only the minimal radiation dose to the reference
target volume tended to have an influence on the LPFR (P = 0.068).
The 2-year LPFRs for patients who
received irradiation doses of ![]() 30 Gy and < 30
Gy were 83% and 52%, respectively. With regard to morbidities, Grade
3 respiratory symptoms (according to the Radiation Therapy Oncology
Group/European Organization for Research and Treatment of Cancer late
radiation morbidity scoring scheme) were noted in one patient.
30 Gy and < 30
Gy were 83% and 52%, respectively. With regard to morbidities, Grade
3 respiratory symptoms (according to the Radiation Therapy Oncology
Group/European Organization for Research and Treatment of Cancer late
radiation morbidity scoring scheme) were noted in one patient.
Single, high-dose irradiation long has been considered prohibitive because of late radiation morbidities. Even as early as the 1920s, German groups published reports of the disastrous effects of single, high-dose irradiation on normal tissues. However, the very confined treatment volumes obtained with SRT has made it possible to apply a single, high dose without causing late morbidities, even in sensitive organs such as the central nervous system. Single-fraction SRT is a promising modality that has the potential to circumvent the problem of repopulation, which can occur during conventional, fractionated radiation therapy, and to improve the local control of lung tumors. However, to our knowledge only a few authors to date have reported results from studies that used single-fraction SRT to lung tumors. Hof reported on 10 patients with histologically confirmed, Stage I, nonsmall cell lung carcinoma who received treatment with single-fraction SRT (isocenter dose range, 19-26 Gy). After 12 months and 24 months, the local recurrence-free survival rate in their study reached 88.9% and 64%, respectively. Wulf reported results from a study of single-dose SRT to 25 lung metastases with a CTV < 25 cc, and no local recurrences were observed at a median follow-up of 9 months (range, 2-37 mos). In that series, the PTV was defined as the CTV plus 5-10 mm margins and was enclosed by 20.8 Gy.
In the current series, all tumors that were
irradiated at a dose < 30 Gy used minimal dose of the CTV, and not the PTV,
as the prescribed dose. Therefore, in some tumors, the minimal dose to the
PTV may have been lower compared with the series reported by Wulf et al. The
poor 2-year LPFR of 52% noted among tumors that were irradiated at a dose <
30 Gy in the current series may have been because of the lower dose.
Conversely, tumors that were irradiated at a minimal dose
![]() 30 Gy demonstrated an LPFR of 83% and, when
considering only tumors that were irradiated at a
dose
30 Gy demonstrated an LPFR of 83% and, when
considering only tumors that were irradiated at a
dose ![]() 30 Gy by using the prescribed dose to the PTV, the 2-year LPFR reached 90%,
a rate that is comparable to the results reported by Wulf and Hof
30 Gy by using the prescribed dose to the PTV, the 2-year LPFR reached 90%,
a rate that is comparable to the results reported by Wulf and Hof
The optimal dose for the control of lung
tumors by single-fraction SRT is unclear. Wulf et al. revealed a
dose-response relation with an advantage for higher doses of SRT in three
fractions. Ohnishi evaluated 245 patients with Stage I nonsmall cell
lung carcinoma who underwent hypofractionated SRT in Japan with a linear
quadratic model and found that patients who received a biologically
effective dose (BED) ![]() 100 Gy at the isocenter
achieved better local control and had better survival rates compared with
patients who received a BED < 100 Gy. However, the linear quadratic model is
not appropriate for radiotherapy with a fractionated dose > 8 Gy. Therefore,
it is difficult to compare single-fraction radiotherapy with multifraction
radiotherapy. Although a statistically significant difference was not
reached, the results of the current study demonstrated that patients who had
tumors that were irradiated at a dose
100 Gy at the isocenter
achieved better local control and had better survival rates compared with
patients who received a BED < 100 Gy. However, the linear quadratic model is
not appropriate for radiotherapy with a fractionated dose > 8 Gy. Therefore,
it is difficult to compare single-fraction radiotherapy with multifraction
radiotherapy. Although a statistically significant difference was not
reached, the results of the current study demonstrated that patients who had
tumors that were irradiated at a dose ![]() 30 Gy
tended to have more favorable outcomes.
Currently, it is preferable to use a
minimal dose of 30 Gy in a single fraction to obtain good local control in
patients who have malignant tumors that measure < 4 cm in greatest
dimension. To our knowledge, it is unknown whether larger doses are
beneficial in improving tumor control further, and the possibility for
incremental dose increases also depends on the morbidities caused by
single-fraction SRT.
30 Gy
tended to have more favorable outcomes.
Currently, it is preferable to use a
minimal dose of 30 Gy in a single fraction to obtain good local control in
patients who have malignant tumors that measure < 4 cm in greatest
dimension. To our knowledge, it is unknown whether larger doses are
beneficial in improving tumor control further, and the possibility for
incremental dose increases also depends on the morbidities caused by
single-fraction SRT.
The
incidence of adverse reactions to single-fraction SRT was very low, as
expected and as reported in other series. The only Grade 3
respiratory morbidities noted in the current series were reported in a
patient who had active tuberculosis and lung fibrosis. However, when an
increase in the single-fraction dose is attempted, even by using
stereotactic, converging X-ray beams, an increment of the dose to the
healthy lung tissue is inevitable. In conventionally fractionated radiation
therapy, a volume ![]() 20 Gy (V20) of irradiated
lung is related closely to the incidence and severity of radiation
pneumonitis. Likewise, we used a volume of irradiated lung
20 Gy (V20) of irradiated
lung is related closely to the incidence and severity of radiation
pneumonitis. Likewise, we used a volume of irradiated lung
![]() Gy (V8) as an indicator of radiation
pneumonitis in single-fraction SRT. It is mandatory to correlate such
indicators with the incidence of adverse reactions to single-fraction SRT in
the treatment of patients with lung tumors.
Gy (V8) as an indicator of radiation
pneumonitis in single-fraction SRT. It is mandatory to correlate such
indicators with the incidence of adverse reactions to single-fraction SRT in
the treatment of patients with lung tumors.
In the current study, patients who had tumors that were irradiated without respiratory gating and with PTV used as the reference volume achieved responses (CR and PR) more frequently, although the nature of the maximal response demonstrated no statistically significant influence on the LPFR. In some patients, the appearance of consolidation around the tumor by radiation pneumonitis may prevent the correct diagnosis of tumor shrinkage, and such patients may be classified erroneously with SD. The limited value of CT diagnosis in radiation response of lung tumors has been reported previously. For the early prediction of local control by SRT, FDG-PET studies may be useful.
The results of the current series demonstrated that single-fraction SRT may be performed safely with excellent local control in patients with lung tumors that measure < 4 cm in greatest dimension. However, longer follow-up will be required to establish the optimal dose with this method CONCLUSIONSThe results from the current study suggested that single-fraction SRT was tolerable and was capable of attaining excellent local control in patients who had malignant lung tumors that measured < 4 cm in greatest dimension.