|
|
 |
 |
 |
 |
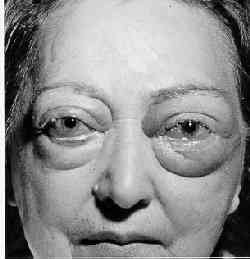
Graves' disease is an autoimmune process (an immunologic response to one's own body tissue/organ) that includes at least one of the following: hyperthyroidism, ophthalmopathy, or infiltrative dermatopathy. Graves' ophthalmopathy (aka: thyroid ophthalmopathy, thyroid-related ophthalmopathy, thyroid eye disease, orbitopathy related to thyroid disorders) is an autoimmune process that is progressive but self-limited with a variable course extending over 1 to 3 years generally and having possible vision-threatening complications. |
Thyroid ophthalmopathy
is clinically apparent in approximately 50% of patients with Graves' disease
and requires intensive treatment or surgical intervention in 3 to 5 percent
of cases. Graves' ophthalmopathy is usually
associated with hyperthyroidism, although patients may be euthyroid (ie. exhibit normal
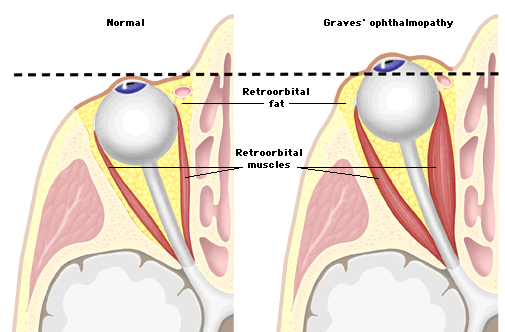
thyroid and hormone levels).Clinical PresentationMild thyroid ophthalmopathy complaints include photophobia (sensitivity of light), foreign body sensation, and increased tearing. Other complaints include discomfort when looking up or restriction of upgaze altogether. Blurred vision from acquired corneal astigmatism (irregularity of the shape of the cornea) is also not unusual. More severe cases may exhibit vertical or horizontal diplopia (double vision). Visual loss may result from corneal ulceration (damage to the cornea) or from optic neuropathy (damage to the optic nerve). (Feldon) "In thyroid ophthalmopathy, a characteristic staring appearance is often present, owing to eyelid retraction. Lid retraction may occur with exophthalmos (abnormal protrusion or bulging forward of the eyeball) or may be an isolated finding. …Early in the course of the disease, this lid retraction may be due to a sympathomimetic (sympathetic nervous system) response, whereas in later stages retraction may be associated with fibrosis (scarring or collagen deposits) of lid tissues. … Patients with thyroid eye disease often complain of excessive lacrimation (tears), gritty sensation, discomfort and photophobia (sensitivity of light)." There may also be conjunctival signs including conjunctival redness and swelling from dilated blood vessels (Albert and Jakobiec, p. 2944-2945). Exophthalmos, also called proptosis, is a characteristic finding in thyroid eye disease, and has been reported to occur in 34% to 93% of patients (Duke-Elder). Abnormalities in the motility of the eyes are common in this disease. Typically the first ocular motility problem is difficult or restricted up-gaze (upward eye rotation) followed later by a limitation of horizontal eye movement. These limitations are often associated with diplopia (double vision). "It should be noted that most thyroid ophthalmopathy patients, including those with no ocular motility symptoms, show some degree of extraocular muscle involvement demonstrated by ultrasonography" (Albert and Jakobiec, p. 2946). Exposure keratitis (corneal inflammation) occurs in these patients and is due to inadequate lid closure due to proptosis and eyelid dysfunction. If left untreated this can result in ulceration of the cornea, perforation, panophthalmitis (inflammation or infection of the entire eye), and loss of the eye. "Visual loss is an important but rare complication of thyroid ophthalmopathy and is usually due to optic neuropathy. The incidence of optic neuropathy is approximately 5% of patients with thyroid ophthalmopathy and affected individuals usually do not have marked proptosis or optic nerve changes on ophthalmoscopy" (Albert and Jakobiec, p. 2947). Ophthalmopathy occurs most frequently in patients with active or treated Graves' disease and in only 2 percent or less of patients with Hashimoto's disease (Wyse, et al.). Rarely, patients with primary hypothyroidism, thyroid cancer, and those with other forms of thyroid disease or thyroid inflammation may have eye findings. (Albert and Jakobiec, p. 2938). Statistics vary, one source states that 10% to 25% of patients presenting with orbital signs and symptoms typical of Graves' disease have no history of thyroid involvement and will test negative serologically (Lemke). Pathology
The mechanism of ophthalmopathy in Graves' disease is controversial. Patients with ophthalmopathy may be hyperthyroid, hypothyroid, or euthyroid; however, most patients who are hypothyroid have been treated for hyperthyroidism and most patients with euthyroid ophthalmopathy have subtle thyroid dysfunction (Jacobson & Gorman.). There is evidence that the ophthalmopathy is a distinct autoimmune disorder separable from Graves' hyperthyroidism (Albert and Jakobiec, p.2938). In Graves' hyperthyroidism, autoantibodies (antibodies to self) bind in the region of the thyroid stimulating hormone [TSH] receptor. This binding causes thyroid hormone production. Some abnormal antibodies bind with the same membrane receptors that bind normal TSH (thyroid stimulating hormone). These antibodies are referred to by several names and acronyms including thyroid stimulating immunoglobulin (TSI), thyroid stimulating antibodies (TSAb), TSH-receptor antibody (TRAb), or possibly Long-Acting Thyroid Stimulator (LATS). These antibodies have a prolonged stimulating effect on the thyroid gland (almost 12 times that for the normal TSH). "The antibodies that cause hyperthyroidism almost certainly develop as the result of autoimmunity that has developed against thyroid tissue. Presumably, at some time in the history of the person, an excess of thyroid cell antigens has been released from the thyroid cells, and this has resulted in the formation of antibodies against the thyroid gland itself" (Guyton, p. 953). It is uncertain whether or not these antibodies are also directed against orbital tissue such as the extraocular muscles or orbital tissue or if a separate set of autoantibodies are circulating. Other possible mechanisms include the role of cell-mediated immunity: "It has been suggested that defects in T-cell subsets (sensitized T-lymphocytes) may promote thyroid ophthalmopathy" (Albert and Jakobiec, p. 2941). The current thought on the mechanism is that "antigens found in orbital tissue are the target of cell-mediated or humoral autoimmune response and subsequent cell damage. The exact nature of the orbital antigen and its relationship to thyroid antigen is unknown." It is also possible that the antigens or T-lymphocytes could directly stimulate such cellular functions as glycosaminoglycan or collagen production (Albert and Jakobiec, p. 2941). DiagnosisDiagnosis is usually made on the basis of the patient's clinical signs and symptoms. However, further tests are clinically useful for determining the type and extent of orbital involvement. The enlarged extraocular muscles can be detected by A-scan or B-scan ultrasonography, or by a CT (computerized tomography) scan. CT scan is especially important when proptosis is not obvious since there is a tendency for apical muscle enlargement in some cases, which can produce compression of the optic nerve. It is also mandatory that patients exhibiting Graves' ophthalmopathy also be examined and assessed for thyroid disease (Feldon). Differential DiagnosisThe ophthalmologist should also consider other types of orbital inflammation as well as neoplastic disease of the orbit: Other causes of bilateral exophthalmos include: Idiopathic inflammatory pseudotumor, Wegener's granulomatosis/vasculitis, metastatic neuroblastoma, leukemia/lymphoma, histiocytosis X, cavernous-sinus thrombosis, congenital orbitofacial malformations. Causes of Chronic unilateral exophthalmos include: pseudotumor, lymphoma, cavernous hemangioma, lacrimal grand tumor, peripheral nerve tumor, meningioma, mucocele, metastatic and secondary tumors (Jones & Jakobiec). Ultrasound or a CT scan usually differentiates these disorders easily (Feldon). TherapyGraves' ophthalmopathy has two broad phases. The first phase is an acute, active inflammatory/congestive one lasting 6 to 18 months (up to 3 years rarely). This phase is believed to be mediated, at least in part, by lymphocytes and is partially responsive to treatment with corticosteroids, immunosuppression, and local radiation therapies. This acute phase is followed by a chronic/stable phase with hypertrophy (increased size) and fibrosis (scarring) of extraocular muscles, lacrimal glands, and orbital fat together with subcutaneous eyelid changes. These later changes are permanent, will not regress or progress spontaneously, and they are unresponsive to any suppressive treatment. Surgical repair is necessary to improve this second phase (Albert and Jakobiec, p.1906). There is no consistent effect of any one type of thyroid treatment on the development, progression, or improvement of ophthalmopathy. Thyroid gland treatment affects only one of the end organs of this disease process and does not alter the fundamental autoimmune process causing orbitopathy. Frequently, in managing these patients, treatment of the thyroid gland must be simultaneous but independent of treatment for ophthalmopathy (Albert and Jakobiec, p. 1905). There is no single simple approach to treating Graves' ophthalmopathy. Each patient presents with a unique set of symptoms and with differing signs which vary in severity. Therapy is tailored to the patient's signs and symptoms and disease present. Treatment may be local, systemic, or surgical. A combination of therapeutic approaches is often required (Feldon). Local or topical therapy is directed at protecting the eyes from exposure and minimizing discomfort while awaiting spontaneous stabilization of the disease process (Albert and Jakobiec, p. 1906). Symptomatic relief can usually be achieved by frequently administering artificial tears, using sunglasses, and taping eyelids closed during sleep. Topical steroids are rarely used and if used the patient should be carefully monitored for corneal ulceration and intraocular hypertension. Ocular exposure is treated with lubricants, moisture chambers, and eyelid taping to maintain the corneal surface. Tarsorrhaphy (stitching the upper and lower eyelids together, partially or completely usually to provide temporary protection to the eye) may be indicated if lubrication becomes a management problem. Lubrication may be tapered or stopped once the active disease phase ceases (Lemke). Systemic drugs have been used to treat different symptoms: Diuretics may help to temporarily decrease periorbital edema. Anti-inflammatory agents, particularly corticosteroids, have been used when ophthalmopathy is progressing rapidly. In the past, immunosuppressive agents such as Azathioprine or cyclophosphamide were used to treat Graves' disease. The success of such treatment is uncertain but it is still occasionally used for recalcitrant cases. (Feldon) "If problems such as significant proptosis, strabismus, or eyelid retraction persist after the disease has become quiescent, surgery is usually indicated. The general sequence of surgical procedures performed in the patient with Graves' disease is orbital decompression, strabismus surgery, and eyelid surgery. The rationale for this sequence is that decompression surgery may affect ocular motility and eyelid position, and strabismus surgery may alter eyelid position. In an individual patient any or none of the three procedures may be indicated" (Lemke, p 3). Optic neuropathy is treated with steroids, radiation, and orbital decompression surgery. High voltage radiation therapy has been used primarily to supplement steroid and surgical treatment of optic neuropathy (Feldon). |
Orbital radiotherapy for Graves' ophthalmopathy. Bartalena L, Marcocci C, Tanda ML, Rocchi R, Mazzi B, Barbesino G, Pinchera A. Thyroid. 2002 Mar;12(3):245-50. Cattedra di Endocrinologia, University of Insubria, Varese, Italy. l.bartalena@libero.it Orbital radiotherapy is a well-established method of treatment for severe Graves' ophthalmopathy, because of its anti-inflammatory and locally immunosuppressive effects. It has been used for 60 years. Conventional external x-ray and cobalt therapy have been abandoned, and most groups now use supervoltage linear accelerators (4-6 MeV). Cumulative doses may vary, but in most studies a cumulative dose of 20 Gy delivered over 2 weeks was utilized. Successful outcome depends on the selection of patients, because recent onset, active ophthalmopathy is much more favorably affected than longstanding, inactive disease. Inflammatory signs, recent onset eye muscle dysfunction, and optic neuropathy respond well to orbital radiotherapy, while proptosis and longstanding eye muscle restriction respond poorly. Overall, favorable responses have been reported, with few exceptions, in approximately 60% of cases. Combination of irradiation with high-dose systemic glucocorticoids provides better results than either treatment alone. Orbital radiotherapy is well tolerated and safe. Preexisting retinopathy (e.g., in patients with diabetes) is a contraindication to this treatment for the risk of further retinal damage. No case of radiation-induced tumors has so far been described after orbital radiotherapy for Graves' ophthalmopathy.
Orbital radiotherapy for Graves' ophthalmopathy: useful
or useless? Safe or dangerous? The aftermath of orbital radiotherapy for
graves' ophthalmopathy. Long-term safety of orbital radiotherapy for
Graves' ophthalmopathy. Long-term results of irradiation for patients
with progressive Graves' ophthalmopathy. Effect of steroid pulse therapy with and
without orbital radiotherapy on Graves' ophthalmopathy.
|