Radiotherapy plus Cetuximab
(Erbitux) for Squamous-Cell Carcinoma of the Head and Neck
James A. Bonner, M.D. Ph.D. NEJM 2006;354:567
The treatment of locoregionally advanced squamous-cell
carcinoma of the head and neck (hereafter called head and neck cancer) has evolved
gradually from surgery as the mainstay of treatment to radiotherapy as the principal
treatment. More recently, additional benefit has been obtained with
altered-fractionation radiotherapy (i.e., accelerated fractionation or hyperfractionated
radiotherapy) and with radiotherapy combined with chemotherapy (chemoradiotherapy).
The value of chemoradiotherapy is, however, counterbalanced by increased and often
prohibitive toxicity, particularly among patients with coexisting medical conditions and
decreased performance status.
The epidermal growth factor receptor (EGFR), a member of the ErbB
family of receptor tyrosine kinases, is abnormally activated in epithelial cancers,
including head and neck cancer. The cells of almost all such neoplasms express
high levels of EGFR, a feature associated with a poor clinical outcome. Radiation
increases the expression of EGFR in cancer cells, and blockade of EGFR signaling
sensitizes cells to the effects of radiation.
Cetuximab (Erbitux, ImClone Systems), an IgG1 monoclonal antibody against the
ligand-binding domain of EGFR, enhances the cytotoxic effects of radiation in
squamous-cell carcinoma. In a preliminary study of radiotherapy plus cetuximab in
patients with locoregionally advanced head and neck cancer, the regimen was well
tolerated, and all the patients who could be assessed had a complete or partial
regression. Cetuximab as a single agent or combined with cisplatin was also associated
with clinically significant rates of tumor regression in patients with platinum-refractory
head and neck cancer. For these reasons, we conducted a randomized, phase 3 study to
determine the effect of adding cetuximab to radiotherapy in the treatment of patients with
locoregionally advanced head and neck cancer. We conducted a multinational,
randomized study to compare radiotherapy alone with radiotherapy plus cetuximab, a
monoclonal antibody against the epidermal growth factor receptor, in the treatment of
locoregionally advanced squamous-cell carcinoma of the head and neck.
Methods Patients with locoregionally advanced head and neck
cancer were randomly assigned to treatment with high-dose radiotherapy alone (213
patients) or high-dose radiotherapy plus weekly cetuximab (211 patients) at an
initial dose of 400 mg per square meter of body-surface area, followed by 250 mg per
square meter weekly for the duration of radiotherapy. The primary end point was the
duration of control of locoregional disease; secondary end points were overall survival,
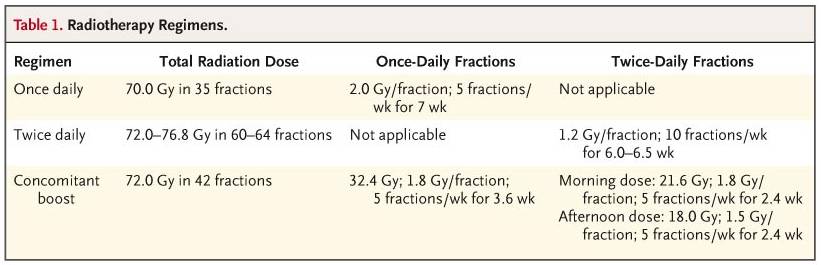
progression-free survival, the response rate, and safety. Regarding the
radiation-fractionation schemes, concomitant boost radiotherapy was selected most
frequently (56 percent), followed by once-daily fractionation (26 percent) and twice-daily
fractionation (18 percent). The final review of radiotherapy revealed that the mean and
median doses for the once-daily, twice-daily, and concomitant-boost regimens were 67.5 and
70.0 Gy, 74.2 and 74.4 Gy, and 71.2 and 72.0 Gy, respectively, with no differences between
the two treatment groups. Compliance was also balanced: overall, 44 percent of the
patients were treated as stipulated, 31 percent received treatment with minor variations,
and 12 percent received treatment with acceptable major variations.
Severe late effects
related to radiation were reported in about 20 percent of the patients in each group.
The sites most commonly affected were the esophagus, salivary glands, larynx, mucous
membranes, subcutaneous tissues, bone, and skin. Twelve patients in the radiotherapy group
and 11 patients in the combined-therapy group died within 60 days after the last
radiotherapy or cetuximab treatment. No death was known to be related to cetuximab.
Results The median duration of locoregional control was 24.4
months among patients treated with cetuximab plus radiotherapy and 14.9 months among those
given radiotherapy alone (hazard ratio for locoregional progression or death,
0.68; P=0.005). With a median follow-up of 54.0 months, the
median duration of overall survival was 49.0 months among patients treated with combined
therapy and 29.3 months among those treated with radiotherapy alone (hazard
ratio for death, 0.74; P=0.03). Radiotherapy plus cetuximab significantly prolonged
progression-free survival (hazard ratio for disease progression or death, 0.70; P=0.006).
With the exception of acneiform rash and infusion reactions, the
incidence of grade 3 or greater toxic effects, including mucositis, did not differ
significantly between the two groups.
Conclusions Treatment of locoregionally advanced head and neck cancer with concomitant
high-dose radiotherapy plus cetuximab improves locoregional control and reduces mortality
without increasing the common toxic effects associated with radiotherapy to the head and
neck. |