|
Cerebral arteriovenous malformations This topic review will discuss cerebral AVMs. Three other general subtypes of congenital vascular malformations have been described, including developmental venous anomalies, capillary telangiectasias, and cavernous malformations.
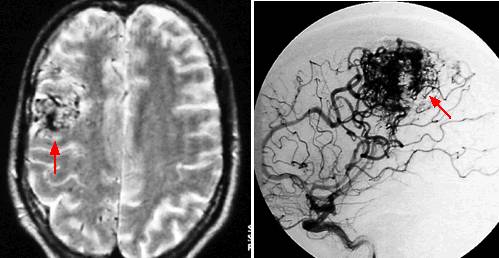
EPIDEMIOLOGY, PATHOGENESIS, AND PATHOLOGY — Intracranial arteriovenous malformations (AVMs) occur in about 0.1 percent of the population, one-tenth the incidence of intracranial aneurysms. Supratentorial lesions account for 90 percent of AVMs; the remainder are in the posterior fossa. AVMs account for 1 to 2 percent of all strokes, 3 percent of strokes in young adults, and 9 percent of subarachnoid hemorrhagesThe pathogenesis of AVMs is not well understood. Genetic variation may influence their development and clinical course. As an example, preliminary evidence suggests that a polymorphism in the inflammatory cytokine IL6 is associated with a three-fold increased risk of ICH among patients harboring AVMs. The angioarchitecture of AVMs is direct arterial to venous connections without an intervening capillary network. Gliotic brain is usually admixed with the vascular tangle, and calcification may be seen in the nidus and surrounding brain. This high-flow arteriovenous communication potentiates a variety of flow-related phenomena such as the development of afferent and efferent pedicle aneurysms and arterialization of the venous limb. Aneurysms can be a source of bleeding in patients with AVMs and are thought to worsen their prognosis The size of AVMs varies widely. There also are a variable number and location of arterial feeders and draining veins that impact upon prognosis and treatment. Regional brain perfusion is compromised due to the AVM "stealing" flow from normal surrounding parenchyma. As a result, histopathological studies demonstrate areas of chronic ischemia and gliosis in the region of the malformation. Arteriovenous malformations (AVMs) usually present in the second to the fourth decade of life. The clinical presentation typically falls into one of three categories: intracranial hemorrhage (41 to 79 percent); seizures (11 to 33 percent); and headaches or progressive deficit. Younger patients (less than 30 years) most often present with seizures, while older patients more commonly present with hemorrhage. A cross-sectional study of 623 prospectively enrolled patients from a tertiary medical center AVM databank found that infratentorial AVM location was independently associated with hemorrhagic presentation.The natural history of unruptured AVMs has been extensively studied. One group followed 168 patients for an average of eight years and reported an annual hemorrhage rate of 2.2 percent, with an average annual morbidity and mortality of 2.8 and 0.7 percent, respectively. Another study reported a somewhat higher rate of bleeding (4 percent per year), with similar rates of major morbidity and mortality The rate of rebleeding is higher during the first year after the initial hemorrhage (6 to 7 percent) and then remains relatively constant. Otherwise, the risk of hemorrhage does not appear to depend upon the manner of presentation Arteriovenous malformations (AVMs) are typically first discovered on cross-sectional imaging (CT or MRI), although angiography is required to plan therapy. A combination of MRI and angiography also is often used to assess the success of surgical, endovascular, or radiosurgical therapy. Flow voids may be identified on CT with contrast administration in and around the region of the nidus of the AVM. CT characteristically demonstrates intraparenchymal hemorrhage without significant edema in patients who present with hemorrhage. However, compression of the nidus by the hematoma often precludes CT diagnosis of AVM in the setting of acute intracerebral hemorrhage; more sensitive techniques such as MRI or angiography are required in such cases. The sensitivity of CT to identify AVMs in the acute setting may be improved by CT angiography. MRI is very sensitive for delineating the location of the nidus and often an associated draining vein. It also has unique sensitivity in demonstrating remote bleeding related to these lesions. Dark flow voids are appreciated on T1 and T2-weighted studiesMRI also is particularly valuable in following patients after treatment. After radiosurgery, for example, the regression of the nidal volume can be accurately measured and followed. In addition, surrounding tissue that is subjected to the radiation field can be accurately monitored for post-therapy edema or, in some cases, radiation necrosis. Angiography is the gold standard for the diagnosis, treatment planning, and follow-up after treatment of AVMs Anatomical and physiological information such as the nidus configuration, its relationship to surrounding vessels, and localization of the draining or efferent portion of the AVM are readily obtained with this technique. Contrast transit times provide additional useful information regarding the flow state of the lesion; this is critical for endovascular treatment planning. A number of variables are factored into the decision to treat cerebral arteriovenous malformations (AVMs) and the therapy chosen. Surgery is the mainstay of treatment; radiosurgery is a useful option in lesions deemed at high risk for surgical therapy, and endovascular embolization has become a useful adjunct to these techniques. Age is probably the most important factor in the decision to treat AVMs since the annual risk of hemorrhage and associated morbidity and mortality statistics are weighed against life expectancy. Thus, children and young adults are usually recommended for therapy, while older individuals with shorter life expectancies may be managed more conservatively.Another important factor in recommending therapy is an assessment of surgical risk. One group used anatomic considerations to construct a classification system (now referred to as the Spetzler-Martin grading scale) that takes into account the lesion size, location, and venous drainage pattern Patients are graded in accordance with these variables; outcomes have consistently been directly related to these factors. High-grade lesions are those of larger size in eloquent brain areas (eg, areas of critical cortical function, deep nuclei, brain stem) with deep venous drainage patterns that make them the most difficult to safely resect. Female gender may also increase surgical risk A simple formula has been devised that considers both age and the risk of morbidity and mortality of treatment versus natural history: P is the annual probability of hemorrhage, and N is the number of years in question. As an example, a 60-year-old female with a newly diagnosed AVM and no other contributing comorbid conditions would expect to live approximately 15 years. Using the formula, the cumulative risk of hemorrhage over her expected life span is 16 percent versus an overall risk of treatment morbidity and mortality of approximately 5 percent, thereby justifying treatment. This calculation only serves as a rough guideline to assist in clinical decision making and is based upon cumulative data rather than an individual patient. Most of the available data suggest that the risk of hemorrhage from an AVM is not increased during pregnancy, but the issue is controversial, and no definitive data exist. One of the better studies was a retrospective analysis of 451 women with an AVM.The AVM hemorrhage rate in pregnant women in this population was not significantly different compared with the rate for nonpregnant women (3.5 versus 3.1 percent per person-year). Stereotactically focused high energy beams of photons or protons to a defined volume containing the AVM nidus induces progressive thrombosis of properly selected lesions via fibrointimal hyperplasia and subsequent luminal obliteration. The time course of these events is usually one to three years, and the time between treatment and obliteration is referred to as the latency period. Once the lesion is completely obliterated, the hemorrhage risk from the AVM is very low, but not totally eliminatedEvidence concerning the hemorrhage risk during the latency period between treatment and obliteration is conflicting. This is an important consideration, particularly for lesions with an aggressive clinical course, such as those presenting with a previous hemorrhage and afferent pedicle or intranidal aneurysms. There are no randomized controlled trials of radiosurgery for AVMs, and none are likely to be performed as the benefit of radiosurgery for radiographic obliteration of AVMs is widely accepted. Existing studies are generally limited by retrospective and observational design. Successful AVM obliteration with radiosurgery depends upon lesion size and dose of radiation. Complete cure is considerably higher with smaller lesions; an overall 80 percent obliteration rate by three years occurs with lesions that are 3 cm or smaller. Larger lesions have reported obliteration rates of 30 to 70 percent at three years. AVMs with a diffuse nidus or associated neovascularity were less likely to achieve a radiographic cure in one clinical series of 248 patients Despite the lower initial success rates for angiographic obliteration seen with larger AVMs (>3 cm), some amount of lesion volume reduction (mean 66 percent) typically occurs, and retreatment is effective for complete obliteration in about 60 percent of patients with residual AVMs. As mentioned above, the success rate of radiographic AVM obliteration varies with the amount of radiation delivered to the margin of the lesion. Doses of 16, 18, and 20 Gy are associated with obliteration rates of about 70, 80, and 90 percent, respectively Although radiosurgery for AVM is usually performed to reduce the lifetime risk of bleeding, improved seizure control may be another benefit for some patients. In a study of 65 patients who had seizures before treatment with stereotactic radiosurgery for AVM, 25 (51 percent) were free of seizures and auras three years after treatment, including the 13 patients who had a single seizure only before treatment. Of the 23 patients with disabling seizures before treatment, 11 (61.1 percent) had an excellent outcome (defined as seizure-free or nondisabling seizures) at three-year follow-up. Better outcomes were associated with a low frequency of seizures before treatment and smaller size AVM. The results of this study are limited by retrospective design and absence of a control group. Both the success rate and incidence of complications are related to the AVM location and the volume treated. Thalamic, basal ganglionic, and brainstem locations are particularly prone to development of deficits after radiosurgery. The risk of complications is also related to the radiation dose directed to the surrounding tissue.The risk of radiation necrosis with permanent neurologic deficit is 2 to 3 percent in most reports. In a multinational study that included 1255 patients undergoing radiosurgery for cerebral AVMs, therapy-related complications developed in 102 (8 percent) and included radiographic parenchymal lesions, cranial nerve deficits, seizures, headaches, and cyst formation. Symptoms were disabling in 21, fatal in two, and resolved completely in 42 (41 percent). In a second series that included 227 patients, only two patients developed treatment-related permanent new neurologic deficits. The increased risk of complications in
large AVMs that require larger treatment volumes was illustrated in a
series of 73 patients, in whom one-half of the AVMs were >3 cm in diameter.
The incidence of post-treatment imaging abnormalities and clinical
complications rose with increasing treatment volume. In patients whose
treatment volumes were >14 mL and who received a dose
In contrast to standard fractionation cranial irradiation, radiosurgery does not appear to impact on cognitive function. One study of 10 patients found no effect of radiosurgical treatment of AVMs upon neuropsychological performance 11 months after treatment. Despite initial optimism that embolic agents such as microparticles and cyanoacrylates could cure AVMs, less than 5 percent of lesions are cured by this approach alone. In addition, flow studies have demonstrated that partial embolization may increase the pedicle pressure and the risk of hemorrhageNevertheless, embolization is an effective adjunct to radiosurgery and surgery. Embolization prior to radiosurgery is employed to reduce the nidus size of large AVMs to less than 10 cm3, as these large AVMs have a lower cure rate with radiosurgery alone. Embolization prior to surgery is employed to reduce blood loss and to occlude vessels that may be difficult to control during surgery. Furthermore, endovascular treatment appears to be associated with a relatively low risk of disabling treatment complications A meticulous analysis of angiographic information determines the suitability for embolization. Generally, only afferent pedicles to the nidus are embolized in an attempt to avoid occlusion of branches irrigating normal brain. Recommendations issued by the American Stroke Association state that surgical extirpation should be considered the primary mode of therapy for low-grade (I and II) lesions based upon the Spetzler-Martin grading scale. Radiosurgery should be considered for small lesions where surgery offers some increased risk based on location or feeding vessel anatomy. Combined treatment with embolization followed by surgery may be used in patients with grade III lesions. Surgery alone in patients with grade IV and V lesions confers a high risk. |